- Research article
- Open access
- Published:
Facile and precise quantitative determination of silicon in naphtha by inductively coupled plasma-optical emission spectroscopy
Journal of Analytical Science and Technology volume 13, Article number: 30 (2022)
Abstract
The amount of silicon in naphtha must be controlled because it results in silica deposits in vehicle components, such as spark plugs, catalytic converters, and oxygen sensors, which can lead to fuel economy degradation, increased emissions, and damage to catalytic converters. Naphtha has low specific gravity and high volatility; plasma does not form during analysis using inductively coupled plasma-optical emission spectrometry. Therefore, the analysis of silicon in naphtha has been conducted using a dilution method with organic solvents. On the other hand, a high dilution factor increases the LOQ (limit of quantitation), making it difficult to analyse below 100 ppb. In this study, propylene glycol monomethyl ether acetate (PGMEA) was used to solve this problem because it has excellent naphtha solubility, low volatility, and is used widely in semiconductor processing. A LOQ and LOD (limit of detection) of up to 3.3 ppb and 1.0 ppb, respectively, could be achieved using a concentration method by diluting naphtha in PGMEA and heating it. In addition, the reference materials were prepared with octamethylcyclotetrasiloxane to ensure analysis accuracy; excellent results were obtained with an 88% recovery rate.
Introduction
Naphtha (CAS NO: 8030-30-6) is a hydrocarbon compound with 4 to 15 carbon atoms that is discharged between the LPG (liquefied petroleum gas) and kerosene fraction when heated to 220°C in the crude oil distillation process (Prestvik et al. 2004). The naphtha used as the main raw material in the petrochemical industry is classified into full-range naphtha, light naphtha, and heavy naphtha. Full- range naphtha belongs to a hydrocarbon component that boils between 30°C and 200°C and accounts for 15-30% of crude oil by mass. Light naphtha boils between 30°C and 90°C and is composed of molecules with 5-6 carbon atoms. Heavy naphtha has a boiling point between 90°C and 200°C and consists of molecules with 6-12 carbon atoms (Melpolder et al. 1952).
Naphtha is used widely in the manufacture of gasoline, ethylene, propylene, and p-xylene through coking. The Si concentration in coked naphtha is generally high, above 1 mgL−1, but very low in cracked naphtha (Gazulla et al. 2017). The silicon in naphtha can result in silica deposits in vehicle components, such as spark plugs, catalytic converters, and oxygen sensors. These silica deposits must be managed because the mixing ratio of fuel and air supplied to the engine cannot be controlled normally, resulting in a decrease in fuel economy, increase in exhaust gases, and damage to the catalytic converter (Understanding the silicon issue 2012). Silica contamination in petroleum refining can have negative effects, including the poisoning of naphtha hydrotreating catalysts (Testing for Silicon in Gasoline and Related Products 2012). When naphtha contaminated with silicon is treated in a hydroprocessor, the silica compound is absorbed on the catalyst surface and proceeds to an irreversible reaction. The desulfurization activity decreases with time, and the life of the catalyst is shortened, making it impossible to regenerate (Britto et al. 2010).
Silicon, which is an important impurity in naphtha, has been determined quantitatively by atomic absorption spectrometry (AAS), inductively coupled plasma-optical emission spectrometry (ICP-OES), and inductively coupled plasma mass spectrometry (ICP-MS). Silicone is present in naphtha as a polysiloxane used as a defoaming agent in many petroleum industry processes. In coke naphtha, silicon is present at high concentrations (> 1.0 µgmL−1) and can be determined easily by flame-AAS using a nitrous oxide-acetylene flame. Chromatography is a method to find a specific species of Si in naphtha, and detect only 50 to 60% of the ICP-OES measurement result. This is because it is impossible to determine all components of Si by chromatography (Chainet et al. 2011). Also XRF is a method used to detect high-concentration components of several tens of ppm or more, it is not suitable for Si determination in ppb units like this experiment (Kadachi and Al‐Eshaikh 2012;Pedrozo-Penafiel et al. 2019).
ICP-MS has very strong and excellent sensitivity in determining the concentration of contaminants in aqueous solutions, but when organic compounds are introduced to the plasma, the physical properties of the plasma completely change due to overload of the plasma from the generation of organic vapour, leading to a decrease in the sensitivity of the equipment. Furthermore, impurities such as coke accumulate in the sampler and skimmer cone, or the plasma is turned off (Kumar 1999; Gazulla et al. 2017). Because ICP equipment recently supplies oxygen gas together with the sample and the organic compound is injected into the analyser after complete combustion in plasma, the effect of the organic compound is greatly reduced. Nevertheless, plasma instability continues to appear in the case of highly volatile materials, such as naphtha.
Silicon has three isotopes of 28Si, 29Si, and 30Si, with relative abundances of 92.23%, 4.47%, and 3.01%, respectively. These Si isotopes are subject to interference by the isobars of 28Si–14N2 and 12C16O+, 29Si–14N15N+, 13C16O+, and 30Si–14N16O+. Hence, the ICP-MS resolution based on a quadrupole cannot solve this isobaric interference, making quantitative analysis difficult (May and Wiedmeyer 1998; Lee et al. 2003; Amais et al. 2013; Wang and Yang 2018).
ICP-OES and ICP-MS are used to determine trace impurities in various fields. On the other hand, the quantitative analysis of trace element impurities by ICP-OES and ICP-MS of some petroleum products and metal alkyls used in the manufacture of semiconductor materials may cause errors. These errors occur due to differences in volatility between the sample for analysis and the standard solution for preparing the calibration curve. To measure organic compounds with a large difference in boiling point by ICP-OES or ICP-MS, when the sample flows from the spray chamber to the torch, a less volatile material is concentrated selectively compared to a material with high volatility. The signal of the volatile sample is higher than that of the non-volatile sample, and the compound having a low boiling point generates a higher signal per mole of the analyte having a high boiling point. Therefore, many errors occur when analysing a sample with different volatility from the standard material used (Al-Ammar et al. 1999). Consequently, matrix matching between the analyte and the standard solution is an important factor in wet analysis.
ICP-OES has been established as a powerful technique for multi-element analysis. On the other hand, the technique can suffer from both spectral and non-spectral interference, limiting the accuracy, repeatability, and reproducibility of the information obtained. Because the nature of the interference is often complex, it is impossible to apply the necessary corrections for an accurate and precise analysis. Traditionally, for quantitative analysis in atomic spectroscopy, a single spectral line free from the criterion for line sensitivity and spectral interference is chosen. Many attempts have been made to correct the spectral interference of ICP-OES, including standard additions, matrix matching, inter-element correction, and optimization of the spectrum baseline selection. Nevertheless, this method has a severe limitation in calibration if the matrix of the analysis sample is complex (Griffiths et al. 2000).
The main drawback after the introduction of ICP-OES was the sample introduction system, which has a significant impact on the analysis performance of the spectrometer. The torch, one of the main areas of hardware that is coupled with the interface, is now made mostly of quartz, a crystal form of SiO2. As the plasma turns on, the temperature gradient increases along the axis. Quartz has a low coefficient of thermal expansion and is relatively resistant to rapid temperature changes, such as when plasma ignition occurs. Because of these two properties, quartz has been used for many years as an ideal material for ICP torches when designed for aqueous sample analysis. On the other hand, when an organic solvent is introduced into the plasma, the temperature gradient along the axis of the ICP torch may increase, resulting in torch devitrification and breakage. The main reason for the increase in the temperature gradient is that high radio-frequency (RF) power is generally used to analyse organic solvents. Carbon-based molecules, the main component of organic solvents, emit a large amount of infrared radiation, which is absorbed by quartz and increases the temperature of the torch (Radial Demountable Ceramic Torch for the Thermo Scientific iCAP 6000 Series ICP spectrometer 2010)
When determining Si in a sample using ICP-OES, torch devitrification causes secondary contamination of Si by SiO2, the main component of the torch, making accurate quantitative analysis difficult. For this reason, the outer tube of a ceramic D-torch, which has been recently applied for various purposes, is made of sialon, a ceramic material derived from silicon nitride, which is durable and stable at high temperatures. The Centre tube is made of alumina, which has excellent chemical and heat resistance properties. Hence, stable analysis can be performed without torch deformation, even in high-temperature plasma. In particular, a D-torch does not contain any SiO2, so there is no problem with the background derived from quartz; it is very powerful in measuring small amounts of silicon.
Many studies have been conducted to analyse impurities in petrochemical products, such as silicon, e.g. naphtha with a low specific gravity and high volatility. Methods for the quantitative analysis of more than tens of ppb have been presented through various types of pretreatment. On the other hand, most of the studies published thus far used two methods, either diluted with a less volatile solvent, such as kerosene, or the solvent is generally cooled before being introduced into the plasma using a cooled spray chamber (Asendorf 2017). These analysis methods have a high likelihood of reduced sensitivity due to the dilution of the sample. Moreover, data errors are likely to occur because the matrix of the naphtha diluted in the solvent and the calibration solvent do not match. In addition, because all of the solvents used in these methods are insoluble in water, an additional oil-based standard solution must be prepared for these experiments, and an expensive cooling device must be installed separately. In this study, propylene glycol monomethyl ether acetate (PGMEA), which is soluble in water and easily diluted in naphtha, was used as a solvent. The conventional method for Si determination was performed by dilution with a highly volatile solvent as mentioned before. The purpose of this study is to overcome the disadvantages of the dilution method to easily and accurately determine Si by trapping it in PGMEA which is non-toxic and has low volatility and high stability against plasma of ICP-OES and selectively evaporating only highly volatile naphtha.
Experimental
Instrument
Thermo Scientific iCAP-7400 ICP-OES was used for silicon determination in naphtha. A ceramic D-torch and 1.0 mm alumina inner tube were used. The spray chamber used a cyclonic spray chamber made of PTFE (polytetrafluoroethylene) material that is resistant to HF and a 400 µl/min PFA (perfluoroalkoxy) microflow nebulizer at room temperature (19°C) without a separate cooling device. The hot plate used for naphtha and PGMEA evaporation was a product of Savillex (USA, HPX-200). The heating work surface area was 292 mm× 212 mm, and was used at 150±2°C. The temperature at the edge is slightly higher than the temperature outside inside the fume hood. The container used for volatilization was prepared by making PFA containing no Si with a height of 40 mm and an area of 490 mm2 and washed with HF solution at 150°C for 24 hours.
Reagents and standard
As a verification method to ensure the accuracy of the analysis results, all procedures, including pretreatment, were performed in the same manner as the certified reference material (CRM), and the analyte and results were compared. On the other hand, a reference material (RM) based on the same matrix can be used if there is no CRM. In this experiment, octanmethylcyclotetrasiloxane (OMCTS, Sigma-Aldrich, CAS No. 556-67-2, density 0.956 g/ml, boiling point 175°C), which was diluted to 200 ppb with the silicon in naphtha to prepare RM, was used. This is one of the siloxanes commonly present in naphtha and causes major silicon contamination problems.
In ICP-MS analysis, the ideal matrix for the sample is water, but naphtha is insoluble in water. Therefore, a 10 ppm mother standard solution was prepared by diluting 1000 ppm Si single standard (AccuStandard, USA) in PGMEA which is soluble in water, and then re-diluted in PGMEA to 10 ppb, 50 ppb, 100 ppb, 200 ppb concentrations to prepare a standard solution for a calibration curve. RM was prepared at 200 ppb by diluting 10 ppm mother solution directly in naphtha.
Naphtha evaporation rate measurement
Naphtha used in the experiment was placed in a clean polyethylene bottle (Aicellomilim, HR013) and stored frozen at − 20°C. Subsequently, 5 g of naphtha was placed into the washed PFA beaker, and 10 g of PGMEA was added. The resulting mixture was heated to 80°C, 100°C, 110°C, 120°C, and 130°C on a hot plate. The weight change was measured at 10-minute intervals for 300 minutes, and the temperature at which all naphtha volatilized was set as the endpoint where the residual liquid became 10 g.
Sample preparation
After 5 g naphtha was placed into a cleaned PFA beaker, 10 g of PGMEA was added, and the hot plate temperature was set to 120°C for 15 g. The amount of residual solution was then recorded at 30 minutes intervals from 90 minutes until 210 minutes. Naphtha samples contaminated with approximately 50 ppb, 100 ppb, and 200 ppb Si were selected to check the accuracy and quantitative limit of the analysis result. The concentration change according to the volatilization time was checked to verify the reliability of the analysis result. Because this work aimed to quantify Si at the ppb level, the silicone concentration did not need to be higher. In addition, to check the loss of silicon components that may occur during the pretreatment process and ensure accuracy, the OMCTS RM sample prepared in 200 ppb was measured in the same manner as the naphtha sample, and 100 ppb samples spiked directly to the naphtha.
Results and discussion
Calibration
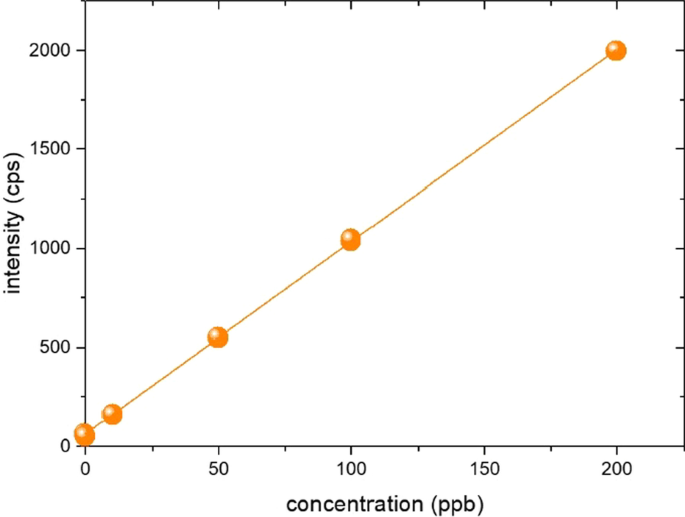
Since naphtha was diluted in PGMEA and all naphtha was volatilized on the hot plate, almost no naphtha was present in the sample for analysis. Therefore, there is no need to consider naphtha in ICP-OES optimization, and most of the final components are PGMEA as shown in Fig. 1. For the optimization of ICP-OES, after preparing PGMEA containing 100 ppb Si to maintain the same matrix as the sample for analysis, the RF power, coolant flow, nebulizer flow, auxiliary flow, and additional gas flow were tested to determine the condition with the largest signal size compared to the equipment background. In ICP-OES, silicon can be measured with two wavelengths of 288.158 nm and 251.611 nm. In this experiment, 251.611 nm with high relative sensitivity was selected (Amais et al. 2013; Gazulla et al. 2017), and the axial view mode was used for detection mode. The test was conducted by varying the RF power from 1150 W to 1500 W. At less than 1350 W, the background did not change, but the sensitivity was lowered, and the background was increased to 1350 W or higher. In the nebulizer, the plasma was unstable at 0.5 L/min or more, so the sensitivity of the equipment was lowered, and the RSD% (relative standard deviation) fluctuated. The ideal signal was obtained at an auxiliary gas flow of 0.5 L/min. If the additional O2 gas is insufficient, a large amount of carbon is formed from the unstable combustion, making the plasma unstable. If too much oxygen is supplied, the plasma is turned off. While checking the plasma light, the amount of O2 gas was determined so that the carbon emission in the plasma was invisible. Table 1 lists the instrumental parameters of the ICP-OES measurement for Si analysis.
The intensity trend of ICP-OES for various Si concentration solutions was identified by quantification using an external calibration method. The results showed that the Si intensity increased with increasing addition of silicon in the PGMEA, and all the samples fell onto a linear line regardless of their variations on the Si concentrations. These results suggest that small variations in the Si concentration during the preparation will not significantly affect the Si determination. Consequently, ICP-OES equipment reads the corresponding data. The linearity (R2), limit of detection (LOD), and limit of quantitation (LOQ) were 1.000, 1.0 ppb, and 3.3 ppb, respectively.
The methodological LOD and LOQ were calculated using the following equation presented by the International Union of Pure and Applied Chemistry (IUPAC) (May and Wiedmeyer 1998; Thomsen et al. 2003; For routine analysis requirements and mid-range sample throughput 2017). The LOD was calculated as an average by measuring the blank value of the blank solution used for calibration and a sample of 100 ppb concentration five times each. The LOD is defined as 10·s where s is value of the standard deviation of the measurements, and the following equation was derived by arranging in more detail by the calculation method suggested by IUPAC, as follows:
where SDblk is the standard deviation of the intensities of the multiple blank measurements. STDconc is the concentration of the standard. STDx is the mean signal for the standard, and BLKx is the mean signal for the blank.
Gazulla et al. diluted naphtha in isooctane to detect a LOD and LOQ of 8 ppb and 25 ppb, respectively. In this study, the Si single standard which were mineral oil based was necessary. The spray chamber temperature had been set to − 9°C because of the high volatility of naphtha and isooctane. But those are not easy to build up in normal labs. In addition, many researchers have proposed an analysis method by diluting in kerosene and xylene, but this has many problems due to the organic matrices. When xylene was introduced to ICP-OES, the plasma increased significantly due to the high volatility, resulting in unstable plasma and poor reproducibility. Toluene use was discontinued owing to its high toxicity (Kumar 1999; Gazulla et al. 2017).
In terms of the precision of the analysis result, the LOD was determined by the size of the standard deviation obtained by repeated measurements of the blank and standard solution. A low LOD means that the standard deviation of the sample is small. The LOD determined by Gazulla et al. was 8 ppb, which was eight times higher than that of 1.0 ppb in this study. In other words, this study made it possible to secure an analysis result that is approximately eight times more precise than the conventional analysis result in terms of sample precision analysis.
Evaporation ratio
After 5 g of naphtha was placed into a washed PFA beaker, 10 g of PGMEA was added and heated to 80°C, 100°C, 110°C, 120°C, and 130°C on a hot plate. The weight change was measured at 10-minute intervals until 300 minutes. The weight change was measured up to 300 minutes at 10-minute intervals. Figure 2 shows that rapid volatilization occurred up to 100 minutes regardless of the temperature. This is because naphtha, which is more volatile than PGMEA, is first volatilized, and the residual naphtha and PGMEA are then volatilized together after 100 minutes. Evaporation at 130℃ can reduce the pretreatment time due to the high evaporation rate, but PGMEA and naphtha volatilize at the same time near the initial boiling temperature of PGMEA (145℃). Hence, the temperature to be applied to the experiment was set to 120℃.
Naphtha analysis results
The silicon concentration of the naphtha sample was measured at intervals of 30 minutes from 90 minutes to 210 minutes on a 120°C hot plate. The data were tabulated. The measured concentration Cmeas is the ICP-OES raw data, and the calculated concentration Ccal is the value converted to the actual concentration using the following equation:
where \(M_{{\text{P}}}^{*}\) is the PGMEA residual amount after volatilization and ΔMn is the amount of naphtha used for volatilization.
After measuring the weight by volatilizing naphtha at 120°C from 90 to 210 minutes at 30 minutes intervals, each concentration was converted using the following conversion equation and plotted in Figs. 3, 4, 5, 6. In ICP-OES analysis, the plasma turned off in the light naphtha where the boiling point was between 30 and 90℃. The boiling point of PGMEA was 146.64℃, which is higher than that of naphtha, and the plasma was maintained stably. When the temperature was kept constant in the naphtha and PGMEA mixture at a ratio of 1:1, naphtha was volatilized first, and PGMEA was volatilized more slowly due to the difference in boiling point. In Fig. 3 the amount of volatilization increased with time, and the measured concentration increased even though the variation pattern of concentration differs depending on the Si concentration because the type of naphtha is different, as explained in the sample preparation part of the Experimental section. The reason for the decrease in concentration sample measured at 150 min in Fig. 3a, c is the nonuniformity of hot plate depending on the sample location inside the fume hood. Because silicon is dissolved in PGMEA, it gradually concentrated, and when converted to the final concentration, all samples showed a constant concentration regardless of the volatilization time. In Fig. 3b, the Cmeas values increase continuously, whereas the Ccal decreases slightly because it is thought Si is also volatized minutely during PGMEA volatilization. From the results, all three samples with silicon concentrations of 50 ppb, 150 ppb, and 200 ppb showed the same trend as the result predicted before the experiment.
The calculated result would not show a constant concentration if a loss of silicon or secondary contamination by the environment occurred during the pretreatment process. The LOD and LOQ are the detection limits using the calibration curve and are arithmetic statistics representing the detection limits of the analysis equipment. In contrast, the method detection limit (MDL) refers to the detection limits in the sample analysis (May and Wiedmeyer 1998; Thomsen et al. 2003; For routine analysis requirements and mid-range sample throughput 2017), i.e. the lowest concentration that can be analysed with an analysis device after the pretreatment. Fig. 3 presents the average concentration range of 83% for the standard 50 ppb silicon and shows an insignificant value at concentrations below 50 ppb, so 50 ppb was selected as the MDL. ICP-OES analysis showed that the measured Si concentration increases with increasing evaporation time, regardless of the Si concentration. When converted to the actual concentration, the ICP-OES background Si intensity did not vary significantly in the evaporation time from 90 to 210 min. Hence, the evaporation temperature and evaporation time did not affect the analysis results, and there were no experimental problems.
Accuracy
Reference material measurement
The reference material was prepared at a Si concentration of 200 ppb using OMCTS to verify the amount of Si in the mixture. A series of analytes with known amounts of reference material were measured and calculated under the same ICP-OES parameters. Table 2 lists the results for the five samples, where the listed Si concentration in each sample is the average of three measurements. The recovery is defined as the ratio of the amount of silicon present in the naphtha sample, which was recovered using the analytical method. It refers to an amount of silicon effectively quantified in relation to the real amount present in the naphtha sample. Table 2 reveals a good recovery rate of 86.07% with an average of 172.14 ppb compared with previous reports (Creed et al. 1994; Griffiths et al. 2000), which can be applied to the results to determine the total concentration of all analytes present in the naphtha samples.
Silicon in naphtha exists as OMCTS, one of the siloxane types, not pure SiO2. The problem is that the calibration curve for ICP-OES analysis was prepared by diluting a standard material made of water-based SiO2 in PGMEA, but the silicon dissolved in PGMEA after naphtha pretreatment is in the form of OMCTS. Therefore, the matrices are different. This can cause large errors in the analysis result. In order to confirm the possibility of this error, two types of samples were prepared. In the first sample, 200 ppb of OMCTS was spiked into naphtha, and in the second sample, water-based silicon was first diluted in PGMEA and then re-diluted in naphtha to prepare 100 ppb of each RM. Because of this test, the silicon content of OMCTS was 37.3%, which caused many errors when diluted to 200 ppb or less. Hence, the reliability of the data could not be secured, so an RM at 200 ppb and water-based samples with a median concentration of 100 ppb were prepared. The pretreatment was carried out in the same manner as the naphtha sample, and the recovery rate of the spiked sample was confirmed after analysis.
Analyses of standard solution spike and naphtha samples
To ensure the accuracy of the analysis results, the concentration analysis method was re-verified after spiking with a concentration of 100 ppb of Si in naphtha. As shown from the spike test results in Fig. 4, the concentration coefficient and Si concentrations increase with increasing evaporation time, similar to pure naphtha. The decrease in concentration of sample measured at 150 min is due to the nonuniformity of hot plate as mentioned earlier. In addition, good recovery rates of 90% to 110% were obtained regardless of the evaporation time.
Table 3 lists the results of the concentration change according to the volatilization time after separating naphtha samples into 10 ppb or less, 50 ppb, 150 ppb, and 200 ppb by Si concentration. In the sample of 10 ppb or less, the average concentration was 5.71 ppb. Even if the volatilization amount changed, it did not deviate significantly from the expected set concentration. The standard deviation range of 41.85 ppb, 133.19 ppb, and 202.66 ppb secured good results of 4-6%. Since the evaluation sample was not an RM sample, the determined value itself is considered the concentration present in the naphtha.
Figure 5 presents the ICP-OES spectrum according to the volatilization time. As the volatilization time evaporates, the peak size also increases in proportion, and both the left and right baselines coincide. This shows that naphtha with a low boiling point was mostly volatilized, and PGMEA solvent mostly existed. If naphtha existed, there would have been a difference in the baseline.
Figure 6 summarizes the change in recovery % according to the volatilization time of OMTCTS and water-soluble Si RM, which are organic base RM materials. As mentioned before, OMCTS was 200 ppb RM, and water-soluble Si RM was 100 ppb. Regardless of the volatilization time, the sample spiked with OMCTS showed a good recovery rate of 86% on average and 96% for the sample spiked with Si in the water base. Hence, the analysis result was not affected by the form of silicon present in the naphtha, organic silicon, or SiO2, even if the volatilization time was as short as 90 minutes. It is also shown that the recovery rate of Si RM is higher than OMCTS RM because the molecular formula of OMCTS is C8H24O4Si4, and it is possible that some OMCTS was lost due to volatilization with naphtha during the pretreatment process. On the other hand, since Si RM is dissolved in water, it is thought that volatilization did not occur.
Conclusions
Many studies have been conducted to find an accurate analysis method of Si present in naphtha using ICP-OES or ICP-MS. Because naphtha is insoluble in water, in most studies, the analysis was performed by diluting in isooctane or kerosene, which has a low specific gravity and boiling point, for Si analysis. However, these methods have disadvantages that a cooling device of the spray chamber is required for ICP analysis, and that an organic-based Si standard is required as a standard material. This work highlighted the advantages of diluting naphtha in PGMEA, which can be purchased easily and enables easy and accurate analysis without a separate pretreatment device. The fact that water-soluble Si standard solution widely used in ICP-OES and ICP-MS can be used without a separate Si standard solution for oil analysis enables accurate analysis in a short time. In particular, plasma stability can be maintained even without cooling of the spray chamber. The recovery rate results showed that the final concentration had a constant value even if the evaporation time was changed. The LOD and LOQ were 1.0 ppb and 3.3 ppb, respectively, and the MDL of the naphtha analysis considering the pretreatment method was 50 ppb, ensuring a stable and excellent analysis method. When naphtha was diluted three times in PGMEA and volatilized at 120°C, reliable results could be obtained at the lowest concentration of 50 ppb. Using OMCTS, RM was prepared and analysed at a concentration of 200 ppb, resulting in good results with a recovery rate of 86%.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Abbreviations
- LOQ:
-
Limit of quantitation
- PGMEA:
-
Propylene glycol monomethyl ether acetate
- LOD:
-
Limit of detection
- LPG:
-
Liquefied petroleum gas
- AAS:
-
Atomic absorption spectrometry
- ICP-OES:
-
Inductively coupled plasma-optical emission spectrometry
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- RF:
-
Radio-frequency
- PTFE:
-
Polytetrafluoroethylene
- CRM:
-
Certified reference material
- RM:
-
Reference material
- OMCTS:
-
Octanmethylcyclotetrasiloxane
- RSD:
-
Relative standard deviation
- R2 :
-
Linearity
- IUPAC:
-
International Union of Pure and Applied Chemistry
- SDblk :
-
The standard deviation of the intensities of the multiple blank measurements
- STDconc :
-
The concentration of the standard
- STDx :
-
The mean signal for the standard
- BLKx :
-
The mean signal for the blank
- C meas :
-
The ICP-OES raw data
- C cal :
-
The calculated concentration Ccal
- \(M_{{\text{P}}}^{*}\) :
-
The PGMEA residual amount after volatilization
- ΔM n :
-
The amount of naphtha used for volatilization
- MDL:
-
The method detection limit
References
Al-Ammar A, Gupta R, Barnes R. Correction for volatility differences between organic sample analytes and standards in organic solutions analyzed by inductively coupled plasma-atomic emission and mass spectrometry. J Anal at Spectrom. 1999;14(5):793–9.
Amais RS, Donati GL, Schiavo D, Nóbrega JA. A simple dilute-and-shoot procedure for Si determination in diesel and biodiesel by microwave-induced plasma optical emission spectrometry. Microchem J. 2013;106:318–22.
Asendorf S. Analysis of trace elements in naphtha using the Thermo Scientific iCAP 7000 Plus Series ICP-OES. 2017. https://assets.thermofisher.com/TFS-Assets/CMD/Application-Notes/AN-43209-ICP-OES-iCAP-7000-Naptha-AN43209-EN.pdf.
Britto JM, Reboucas MV, Bessa I. Troubleshoot silicon contamination on catalysts. Hydrocarb. Process.2010. p. 65–69.
Chainet F, Courtiade M, Lienemann C-P, Ponthus J, Donard OFX. Silicon speciation by gas chromatography coupled to mass spectrometry in gasolines. J Chromatogr A. 2011;1218(51):9269–78.
Creed J, Brockhoff C, Martin T. Method 200.8, Revision 5.4: Determination of Trace Elements in Waters and Wastes by Inductively Coupled Plasma-Mass Spectrometry in Development, USEPA-O. o. R. a., ed. 1994;5:58.
For routine analysis requirements and mid-range sample throughput. 2017. https://assets.thermofisher.com/TFS-Assets/CMD/Specification-Sheets/PS-43230-ICP-OES-iCAP-7400-PS43230-EN.pdf.
Gazulla M, Rodrigo M, Orduña M, Ventura M, Andreu C. High precision measurement of silicon in naphthas by ICP-OES using isooctane as diluent. Talanta. 2017;164:563–9.
Griffiths ML, Svozil D, Worsfold PJ, Denham S, Evans EH. Comparison of traditional and multivariate calibration techniques applied to complex matrices using inductively coupled plasma atomic emission spectroscopy. J Anal at Spectrom. 2000;15(8):967–72.
Kadachi AN, Al-Eshaikh MA. Limits of detection in XRF spectroscopy. X-Ray Spectrom. 2012;41(5):350–4.
Kumar S. Determination of trace elements in naphtha by inductively coupled plasma mass spectrometry using water-in-oil emulsions. J Anal at Spectrom. 1999;14(6):967–71.
Lee LY, Fassett JD, Lindstrom AP. Determination of Si in standard reference material SRM 295x Silica-on-Filter. J Anal at Spectrom. 2003;18(7):738–41.
May TW, Wiedmeyer RH. A table of polyatomic interferences in ICP-MS. At Spectrosc. 1998;19:150–5.
Melpolder F, Brown R, Young W, Headington C. Composition of naphtha from fluid catalytic cracking. Ind Eng Chem. 1952;44(5):1142–6.
Pedrozo-Penafiel MJ, Doyle A, Mendes LAN, Tristao MLB, Saavedra Á, Aucelio RQ. Methods for the determination of silicon and aluminum in fuel oils and in crude oils by X-ray fluorescence spectrometry. Fuel. 2019;243:493–500.
Prestvik R, Moljord K, Grande K, Holmen A. Compositional analysis of naphtha and reformate. In: Antos G, Aitani A, editors. Catalytic naphtha reforming. New York: CRC Press; 2004. p. 1–34.
Testing for Silicon in Gasoline and Related Products. 2012. https://sn.astm.org/?q=update/testing-silicon-gasoline-and-related-products-so12.html.
Thomsen V, Schatzlein D, Mercuro D. Limits of detection in spectroscopy. Spectroscopy. 2003;18(12):112–4.
Radial Demountable Ceramic Torch for the Thermo Scientific iCAP 6000 Series ICP spectrometer. 2010. http://tools.thermofisher.com/content/sfs/brochures/D01563~.pdf.
Understanding the silicon issue. 2012. https://www.xos.com/landing-pages/understanding-silicon-contamination.
Wang Q, Yang Z. Quantification of Si in silicone oils by ICP-OES. SILICON. 2018;10(1):1–10.
Acknowledgements
This study was supported by the Academic Research Fund of Hoseo University in 2019 (2019-0824).
Funding
This work was supported by the Academic Research Fund of Hoseo University in 2019 (2019-0824).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by DS and SJ. The first draft of the manuscript was written by SJ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
There are no competing interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Joo, S., Suh, D. Facile and precise quantitative determination of silicon in naphtha by inductively coupled plasma-optical emission spectroscopy. J Anal Sci Technol 13, 30 (2022). https://doi.org/10.1186/s40543-022-00339-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40543-022-00339-0